Leading the way out of opiate addiction
NALMEFENE CONSTA® (SmartDepot – Nalmefene extended-release injection)
NALMEFENE CONSTA® must be administered under the supervision of a physician.
HIGHLIGHTS OF PRESCRIBING INFORMATION
NALMEFENE CONSTA® (Nalmefene extended-release injection) for intramuscular use
NALMEFENE CONSTA® should be used in conjunction with counseling and support.
INDICATIONS AND USAGE
NALMEFENE CONSTA® is an opioid antagonist indicated for the treatment of Opioid Dependence. Opioid-dependent patients must be opioid-free at the time of initial NALMEFENE CONSTA® administration.
NALMEFENE CONSTA® is indicated for the prevention of relapse to opioid dependence, following opioid detoxification.
NALMEFENE CONSTA® should be part of a comprehensive management program that includes psychosocial support.
NALMEFENE CONSTA® is not a substitute for emergency medical care.
DOSAGE AND ADMINISTRATION
Due to different release characteristics, the dosage strengths are not additive and must be selected based upon the desired dosing schedule.
NALMEFENE CONSTA® 393.1 mg for 3-month administration, given as a single intramuscular injection every 12 weeks.
NALMEFENE CONSTA® 786.2 mg for 6-month administration, given as a single intramuscular injection every 24 weeks.
NALMEFENE CONSTA® 1572.4 mg for 12-month administration, given as a single intramuscular injection every 48 weeks.
The injection should be administered by a healthcare professional as an intramuscular (IM) gluteal injection, alternating buttocks for each subsequent injection.
NALMEFENE CONSTA® must not be administered intravenously or subcutaneously (Avoid inadvertent administration into a blood vessel).
DOSAGE FORMS AND STRENGTHS
Injection: 393.1 mg, 786.2 mg, and 1572.4 mg Nalmefene solution in a microsphere formulation (polyLactide-co- glycolide).
CONTRAINDICATIONS
Patients known to be hypersensitive to Nalmefene. Patients with acute hepatitis or liver failure. Patients receiving opioid analgesics. Patients with current physiologic opioid dependence. Patients in acute opioid withdrawal. Any individual who has failed the Nalmefene challenge test or has a positive urine screen for opioids . Patients who have previously exhibited hypersensitivity to Nalmefene, polylactide-co- glycolide (PLG).
WARNINGS AND PRECAUTIONS
Due to the duration of action, keep the patient under continued surveillance Use in patients who are opioid dependent may precipitate acute abstinence syndrome (opioid withdrawal may be life-threatening if not recognized and properly treated).
ADVERSE REACTIONS
The following adverse reactions have been identified during use of Nalmefene: Hypotension, hypertension, ventricular tachycardia and fibrillation, dyspnea. Abrupt reversal of opioid effects in persons who were physically dependent on opioids has precipitated signs and symptoms of opioid withdrawal including: body aches, fever, sweating, runny nose, sneezing, piloerection, yawning, weakness, shivering or trembling, nervousness, restlessness or irritability, diarrhea, nausea or vomiting, abdominal cramps, increased blood pressure, tachycardia. These are not all side effects of the drug. This is only a summary of the most important information about the medicine. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. You are encouraged to report all side effects to the AURUM Group. Visit www.aurumgroup.org.uk or call +447535409827
DRUG INTERACTIONS
Nalmefene antagonizes the effects of opioid-containing medicines, such as cough and cold remedies, antidiarrheal preparations and opioid analgesics.
DESCRIPTION
NALMEFENE CONSTA® (Nalmefene extended-release injection) for intramuscular use is supplied as a microsphere formulation of Nalmefene. Nalmefene is an opioid antagonist with little, if any, opioid agonist activity.
Nalmefene is chemically designated 17-cyclopropylmethyl-4,5α-epoxy-6-methylenemorphinan-3,14-diol
The molecular formula is C21H25NO3
The structural formula is:
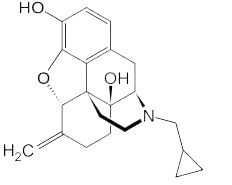
Nalmefene is practically insoluble in water, freely soluble in methylene chloride, and soluble in methanol and 0.1 N HCl. Long-Acting Injection is a combination of extended-release microspheres for injection and diluent for parenteral use. Long-Acting Injection is provided as a dose pack, consisting of a vial containing Nalmefene microspheres and diluent.
The extended-release microspheres formulation is a white to off-white , free-flowing powder that is available in dosage strengths of 393.1 mg, 786.2 mg, or 1572.4 mg Nalmefene per vial. Nalmefene is micro-encapsulated in 5047-14754 polylactide-co- glycolide (PLG) at a concentration of 367.7 mg Nalmefene per 100 mg of microspheres. The diluent for parenteral use is a clear, colorless solution. Composition of the diluent includes citric acid anhydrous, disodium hydrogen phosphate dihydrate, polysorbate 20, sodium carboxymethyl cellulose, sodium chloride, sodium hydroxide, and water for injection or pure solution of calcium gluconate. The microspheres must be suspended in the diluent prior to injection.
SUPPLYING / STORAGE AND HANDLING
NALMEFENE CONSTA® is available in dosage strengths of 393.1 mg, 786.2 mg, or 1572.4 mg Nalmefene. It is provided as a dose pack, consisting of a vial containing the nalmefene microspheres, diluent, 3 pills for Nalmefene challenge test, Medication Guide and Directions for Use.
393.1- mg vial/kit (ATC N07BB05-05P): 500 mg (equivalent to 393.1 mg of nalmefene) of a white to off-white powder provided in a vial with a rubber stopper in combination with aluminum and polypropylene cap. Each pack contains 3 vials.
786.2-mg vial/kit (ATC N07BB05-10P): 10000 mg (equivalent to 786.2 mg of nalmefene) of a white to off-white powder provided in a vial with a rubber stopper in combination with aluminum and polypropylene cap. Each pack contains 3 vials.
1572.4 -mg vial/kit (ATC N07BB05-20P): 2000 mg (equivalent to 1572.4 mg of nalmefene) of a white to off-white powder provided in a vial with a rubber stopper in combination with aluminum and polypropylene cap. Each pack contains 3 vials.
Storage and Handling
The entire dose pack should be stored in the refrigerator (36°-46°F; 2°-8°C) and protected from light. If refrigeration is unavailable, can be stored at temperatures not exceeding 77°F (25°C) for no more than 7 days prior to administration. Do not expose unrefrigerated product to temperatures above 77°F (25°C)
Manufactured by:
Nalmefene active ingredient is manufactured by: Lundbeck A/S, Valby, Denmark.
Microspheres are manufactured by: Peptron, Inc. Biotechnology , Yuseong-gu, Daejeon, Rep. of KOREA.
Diluent is manufactured by: Vetter Pharma Fertigung GmbH & Co. KG. Langenargen, Germany or by S.A.I.F. S.p.a. Laboratorio Farmacologico, Cenate Sotto (Bergamo), Italy.
Manufactured for Aurum Pharmaceutical Ltd
(Provided for research and clinical use only. Not for distribution or commercial use.)
ESPL No: 12064/0066-1/2016